
- Gas Ideal Monoatomik Sebanyak 0 5 Kmol
- Gas Ideal Monoatomik Adalah
- Sejumlah Gas Ideal Monoatomik Mula Mula
- Gas Ideal Monoatomik Memiliki Derajat Kebebasan
Suatu gas ideal monoatomik di dalam ruang tertutup mempunyai tekanan 1,2 x 10 5 Pa dan volume 40 liter. Bila gas memuai secara isobarik sehingga volumenya menjadi 50 liter, maka gas akan menyerap kalor dari lingkungan sebesar 2 x 10 3 J. Pada akhir proses. (1) Suhu gas akan bertambah (2) Gas melakukan usaha sebesar 1,2 x 10 3 J. Ideal gas for 1-, 2-, and 3-D case. Case Partition function 1-D 1/2 2 (1) 2 = h mkT Z L π 2-D 2/2 2 (2) 2 = h mkT Z A π 3-D 3/2 2 (3) 2 = h mkT Z V π NkT We can then say that the partition function of monatomic ideal gas can be written in general form, which is /2 2 ( ) 2 D D D h mkT Z L = π, (23) where D, which is the dimension, can have value of 1, 2, or 3.
Gas Ideal Monoatomik Sebanyak 0 5 Kmol
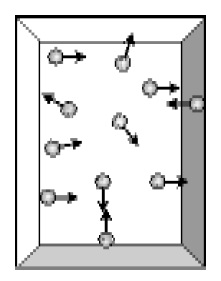
In physics and chemistry, 'monatomic' is a combination of the words 'mono' and 'atomic', and means 'single atom'. It is usually applied to gases: a monatomic gas is one in which atoms are not bound to each other. Examples at standard conditions include the noble gases argon, krypton, and xenon, though all chemical elements will be monatomic in the gas phase at sufficiently high temperatures. The thermodynamic behavior of a monatomic gas is extremely simple when compared to polyatomic gases because it is free of any rotational or vibrational energy.[1]
Gas Ideal Monoatomik Adalah
Noble gases[edit]
The only chemical elements that are stable single atommolecules at standard temperature and pressure (STP) are the noble gases. These are helium, neon, argon, krypton, xenon, and radon. Noble gases have a full outer valence shell making them rather non-reactive species.[2] While these elements have been described historically as completely inert, chemical compounds have been synthesized with all but neon and helium.[3]
When grouped together with the homonucleardiatomic gases such as nitrogen (N2 Sonic 3 android. ), the noble gases are called 'elemental gases' or 'molecular gases' to distinguish them from molecules that are also chemical compounds.
Thermodynamic properties[edit]
Sejumlah Gas Ideal Monoatomik Mula Mula
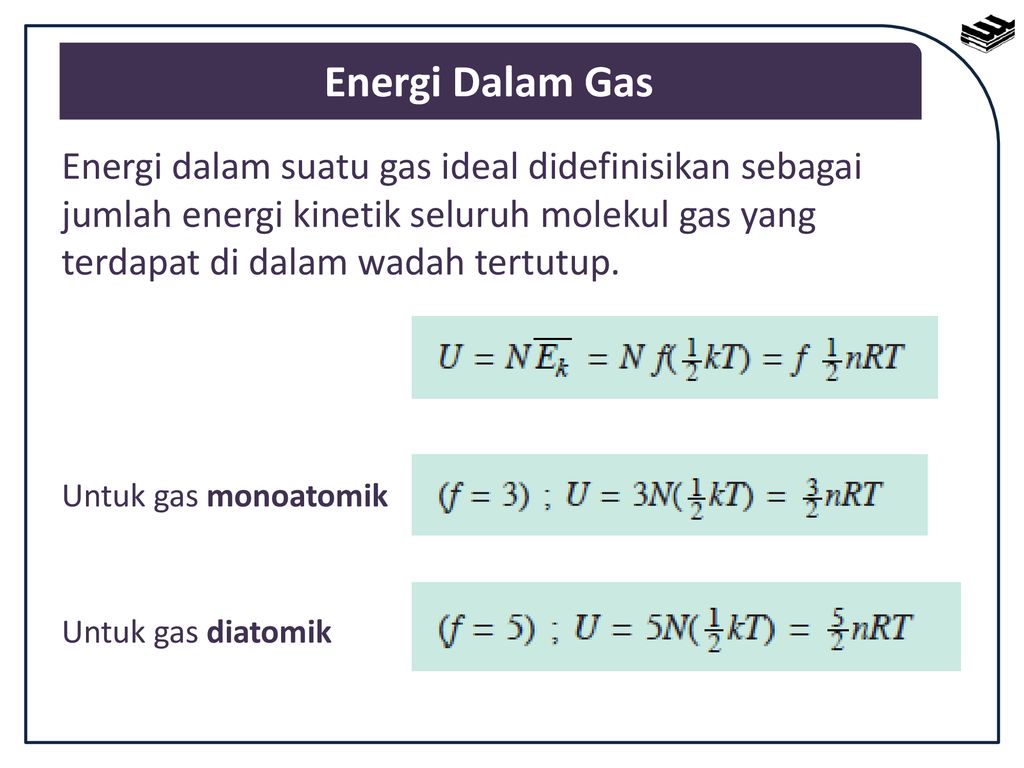
The only possible motion of an atom in a monatomic gas is translation (electronic excitation is not important at room temperature). Thus by the equipartition theorem, the kinetic energy of a single atom of a monatomic gas at thermodynamic temperatureT is given by 32kbT{displaystyle {frac {3}{2}}k_{b}T}, where kb is Boltzmann's constant. One mole of atoms contains an Avogadro number (Na{displaystyle {mathcal {N}}_{a}}) of atoms, so that the energy of one mole of atoms of a monoatomic gas is 32kbTNa=32RT{displaystyle {frac {3}{2}}k_{b}T{mathcal {N}}_{a}={frac {3}{2}}RT}, where R is the gas constant.
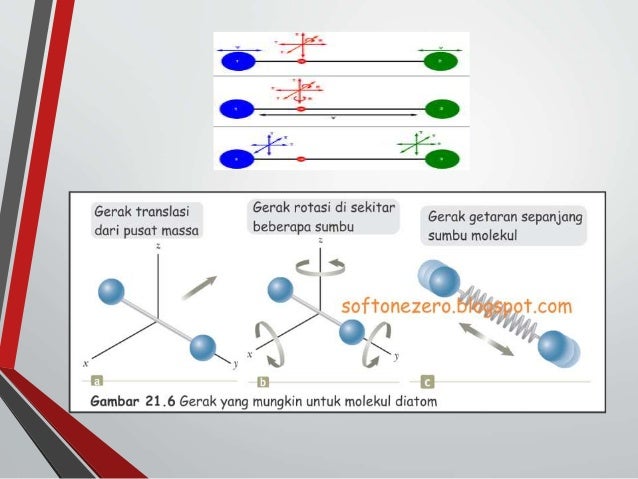
In an adiabatic process, monatomic gases have an idealised γ-factor (Cp/Cv) of 5/3, as opposed to 7/5 for ideal diatomic gases where rotation (but not vibration at room temperature) also contributes. Also, for ideal monatomic gases:[4][5][6]

- Gas Ideal Monoatomik Sebanyak 0 5 Kmol
- Gas Ideal Monoatomik Adalah
- Sejumlah Gas Ideal Monoatomik Mula Mula
- Gas Ideal Monoatomik Memiliki Derajat Kebebasan
Suatu gas ideal monoatomik di dalam ruang tertutup mempunyai tekanan 1,2 x 10 5 Pa dan volume 40 liter. Bila gas memuai secara isobarik sehingga volumenya menjadi 50 liter, maka gas akan menyerap kalor dari lingkungan sebesar 2 x 10 3 J. Pada akhir proses. (1) Suhu gas akan bertambah (2) Gas melakukan usaha sebesar 1,2 x 10 3 J. Ideal gas for 1-, 2-, and 3-D case. Case Partition function 1-D 1/2 2 (1) 2 = h mkT Z L π 2-D 2/2 2 (2) 2 = h mkT Z A π 3-D 3/2 2 (3) 2 = h mkT Z V π NkT We can then say that the partition function of monatomic ideal gas can be written in general form, which is /2 2 ( ) 2 D D D h mkT Z L = π, (23) where D, which is the dimension, can have value of 1, 2, or 3.
Gas Ideal Monoatomik Sebanyak 0 5 Kmol
In physics and chemistry, 'monatomic' is a combination of the words 'mono' and 'atomic', and means 'single atom'. It is usually applied to gases: a monatomic gas is one in which atoms are not bound to each other. Examples at standard conditions include the noble gases argon, krypton, and xenon, though all chemical elements will be monatomic in the gas phase at sufficiently high temperatures. The thermodynamic behavior of a monatomic gas is extremely simple when compared to polyatomic gases because it is free of any rotational or vibrational energy.[1]
Gas Ideal Monoatomik Adalah
Noble gases[edit]
The only chemical elements that are stable single atommolecules at standard temperature and pressure (STP) are the noble gases. These are helium, neon, argon, krypton, xenon, and radon. Noble gases have a full outer valence shell making them rather non-reactive species.[2] While these elements have been described historically as completely inert, chemical compounds have been synthesized with all but neon and helium.[3]
When grouped together with the homonucleardiatomic gases such as nitrogen (N2 Sonic 3 android. ), the noble gases are called 'elemental gases' or 'molecular gases' to distinguish them from molecules that are also chemical compounds.
Thermodynamic properties[edit]
Sejumlah Gas Ideal Monoatomik Mula Mula
The only possible motion of an atom in a monatomic gas is translation (electronic excitation is not important at room temperature). Thus by the equipartition theorem, the kinetic energy of a single atom of a monatomic gas at thermodynamic temperatureT is given by 32kbT{displaystyle {frac {3}{2}}k_{b}T}, where kb is Boltzmann's constant. One mole of atoms contains an Avogadro number (Na{displaystyle {mathcal {N}}_{a}}) of atoms, so that the energy of one mole of atoms of a monoatomic gas is 32kbTNa=32RT{displaystyle {frac {3}{2}}k_{b}T{mathcal {N}}_{a}={frac {3}{2}}RT}, where R is the gas constant.
In an adiabatic process, monatomic gases have an idealised γ-factor (Cp/Cv) of 5/3, as opposed to 7/5 for ideal diatomic gases where rotation (but not vibration at room temperature) also contributes. Also, for ideal monatomic gases:[4][5][6]
- the molar heat capacity at constant pressure (Cp) is 5/2 R = 20.8 J K−1 mol−1 (4.97 cal K−1 mol−1).
- the molar heat capacity at constant volume (Cv) is 3/2 R = 12.5 J K−1 mol−1 (2.98 cal K−1 mol−1).
References[edit]
Gas Ideal Monoatomik Memiliki Derajat Kebebasan
- ^'monatomic gas'. Encyclopædia Britannica. Retrieved 6 June 2016.
- ^Laszlo, Pierre; Schrobilgen, Gary J. (1988-04-01). 'Ein Pionier oder mehrere Pioniere? Die Entdeckung der Edelgas-Verbindungen'. Angewandte Chemie. 100 (4): 495–506. doi:10.1002/ange.19881000406. ISSN1521-3757.
- ^Christe, Karl O. (2001-04-17). 'A Renaissance in Noble Gas Chemistry'. Angewandte Chemie International Edition. 40 (8): 1419–1421. doi:10.1002/1521-3773(20010417)40:8<1419::aid-anie1419>3.0.co;2-j. ISSN1521-3773. PMID11317290.
- ^Heat Capacity of an Ideal Gas
- ^Heat Capacity of Ideal Gases
- ^Lecture 3: Thermodynamics of Ideal Gases & Calorimetry[permanent dead link], p. 2
